The central theme of the lab is postnatal or adult hippocampal neurogenesis (AHN), which is implicated in memory formation and mood regulation. We investigate environmental and molecular regulatory mechanisms controlling AHN by using in vitro approaches (human hippocampal stem cells) for molecular focused studies and mice as animal models for behavioural centred investigations.
We study AHN (i) in a healthy context - such as the effect of diet or the changes occurring upon healthy ageing but also (ii) in the context of diseases such as Alzheimer’s disease and depression. By approaching AHN in the context of health and disease, the strategy is two folds: (i) validating AHN as a target for prevention and pharmacological interventions as well as (ii) developing AHN as a biomarker of disease prediction and progression.
Lab description
The adult mammalian brain contains small populations of neural stem cells dividing and differentiating into neurons. This process of neurogenesis occurs in the subgranular zone of the hippocampal dentate gyrus. AHN decreases with age and elderly animals display impaired learning and memory abilities, whereas increased AHN is linked to improved learning and memory. Moreover, AHN is reduced by stressful experiences and in animal models of depression, whereas many antidepressant treatments have been shown to enhance and are dependent on functional AHN. Therefore, AHN strongly emerges as an advantageous target for counteracting the effect of ageing and stress and thus preventing cognitive and mood decline.
In the Thuret lab we investigate the molecular mechanisms governing neurogenesis by using human hippocampal cell lines and we use the mouse as animal model to study cognition and mood. We perform a wide range of molecular biology and histology techniques with both in vivo and in vitro models. We are currently studying the molecular mechanisms by which environment (i.e. diet) and diseases (i.e. Alzheimer’s Disease) impacts on AHN and subsequently affect cognition and mood.
Research projects
Alzheimer’s Disease and Healthy Ageing
We study how the systemic milieu modulates hippocampal neurogenesis during ageing and Alzheimer’s disease progression.
Depression
We have developed an in vitro model of stress using human hippocampal progenitor cells. We use this model to study the preventive and treatment mode of action of compounds on decreased neurogenesis induced by stress. We are studying the role of the systemic milieu, inflammation and stress in mediating the changes in neurogenesis in depression.
Nutrition
We study the molecular mechanisms (including epigenetic changes and modification in microRNA expression) by which diet content and meal frequency impacts on adult hippocampal neurogenesis and subsequently affects cognition and mood.
We are also investigating if certain bioactives previously shown to have beneficial effect on cognition and mood can regulate adult hippocampal neurogenesis. Furthermore we are studying if their effect on cognition and mood is mediated by changes in neurogenesis.
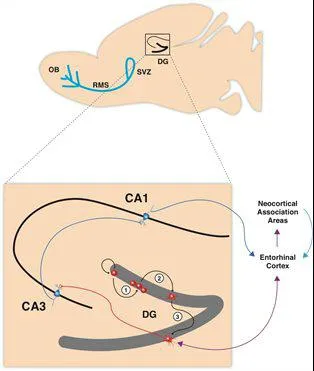
Fig.1: Schematic representation of the sagittal view of a rodent brain highlighting the two neurogenic zones of the adult mammalian brain: The subventricular zone (SVZ) of the lateral ventricles and the subgranular zone of the dentate gyrus (DG) in the hippocampus. Neurons generated in the SVZ migrate through the rostral migratory stream (RMS) and are incorporated into the olfactory bulb. The hippocampal region contained in the black square is enlarged showing (1) neural progenitor cells in the in the subgranular zone of the dentate gyrus proliferating, (2) migrating into the granule cell layer and (3) maturing into new granule neurons, integrating into the hippocampal circuitry by receiving inputs from the entorhinal cortex, and extend projections into the CA3 (Stangl & Thuret, 2009).
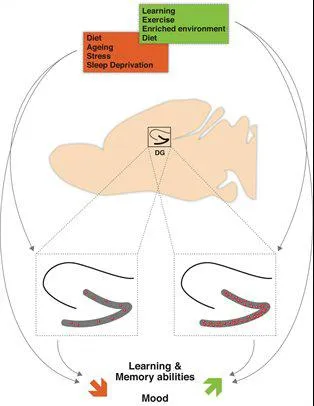
Fig.2: Overview of physiological and environmental modulation of adult hippocampal neurogenesis and its impact on learning and memory abilities and mood. The doted squares contain the enlarged hippocampus. The red dots symbolize newborn neurons in the dentate gyrus (DG) (Stangl & Thuret, 2009)
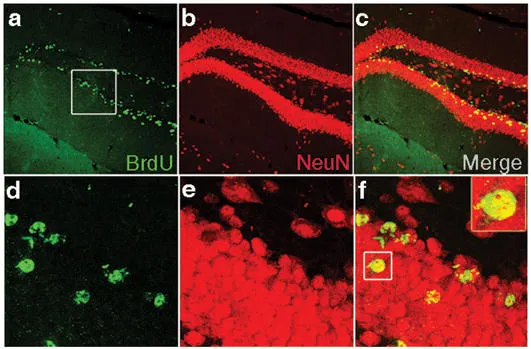
Detection of proliferation and neurogenesis in the dentate gyrus of C57BL/6 mice. Confocal images of dividing BrdU-positive cells in the dentate gyrus. Sagittal sections were imuunostained and double labelled for BrdU (green) and the neuronal marker NeuN (red). New-born/dividing neurons appear in yellow. d, e, f are higher magnification of the boxed area in a, b, c respectively (from Thuret et. al, 2009).
Can we grow new brain cells?
Can we, as adults, grow new neurons? Neuroscientist Sandrine Thuret says that we can, and she offers research and practical advice on how we can help our brains better perform neurogenesis - improving mood, increasing memory formation and preventing the decline associated with aging along the way (credit Ted.com).
