
King's Health Partners
An Academic Health Sciences Centre for London
King’s looks outwards and strongly values its local, national and international research partnerships. External collaborations are a key part of many of our leading research programmes, our multidisciplinary institutes and centres of excellence. This page presents King’s major corporate relationships with key London and national partners, each of which connects with several faculties. Our many other research partnerships are shown on the pages of our departments, institutes, and centres. Our large doctoral training programmes are built on partnerships with other leading universities and with research institutes, offering our students access to a range of networks across the research landscape.


An Academic Health Sciences Centre for London

King’s is one of three founding universities in the Francis Crick Institute

Developing new technologies to tackle important health research challenges

King’s is part of the London Hub of Health Data Research UK

A multidisciplinary enterprise operating at the forefront of science and...
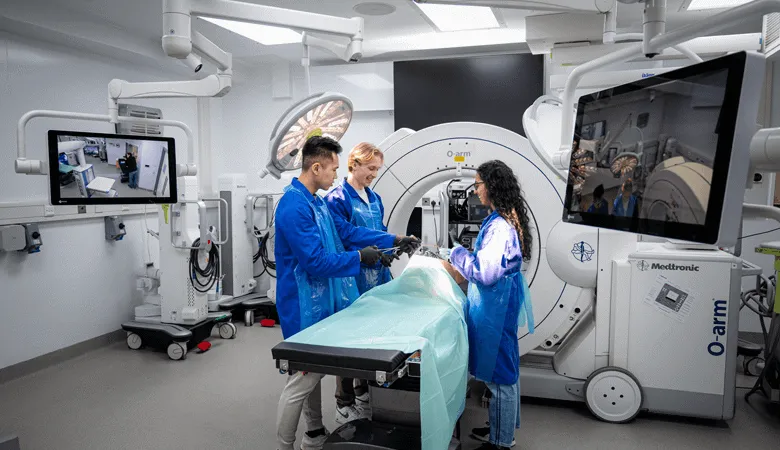
Cutting-edge facilities, technical services and technology platforms.

We offer a streamlined service for commercial sponsors wishing to conduct...

Collaborate with King's on a research project in London.

King's has over 300 partnership agreements with leading institutions...

Your gateway to innovation at King’s
Explore licensing and investment opportunities from King’s researchers.

Get support with initiating collaborations with King’s.
Our students are offered a wider range of research projects and learning opportunities, while also building new networks of connections across the research landscape.
Doctoral training entities