16 December 2019
Novel method to identify tumours with AI in development by School researchers
Artificial Intelligence algorithms to provide surgeons with greater accuracy to delineate tumours
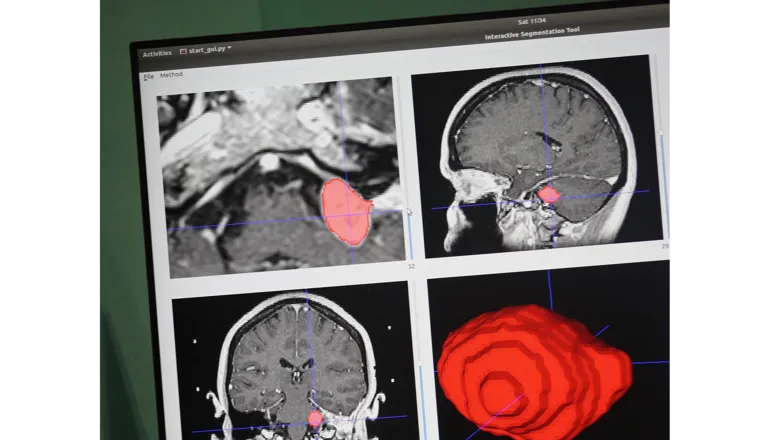
Researchers at the School of Biomedical Engineering & Imaging Sciences at King’s College London, in collaboration with University College London are developing a fully automated method to delineate vestibular schwannoma, a type of brain tumour, with a novel method that can be used to provide treatment and to determine the size of a tumour quicker and more accurately. Results have been shown to be equal and comparable to that of an experienced clinician.
Currently, when identifying a tumour in a medical image, clinicians need to create a tumour model or volume by drawing around each slice of a medical image individually. The only available software to do this is semi-automated which still requires a high level of user interaction, making it more time consuming and less accurate.
“As our software automates the process of identifying where the tumour is in the image and then delineating and segmenting the tumour automatically, the user doesn’t have to do anything other than give the program the image, and click ‘go’ to generate results,” lead researcher Mr Jonathan Shapey said.
The tool allows for the automatic segmentation of the tumour by identifying where the tumour is within the image and drawing around it to delineate the tumour’s border.
After this, the software allows the clinician to change, correct and improve the segmentation to ensure it is as accurate as possible.
With this software tumours can be monitored more effectively, allowing clinicians to get a better idea of size and if whether the tumours are growing or not.
Mr Shapey said the unique interactive feature of the tool is what makes it as accurate as possible.
“If it has identified part of the image as tumour when it is not, the user can correct this,” he said.
At the moment, tumours are measured in a single, linear dimension based on the largest tumour image but small changes in linear dimensions can result in larger volumetric dimensions.
It has already been shown that volumetric measurements are better at documenting size of tumour and detecting subtle growth in the tumour.
By giving clinicians a tool to be able to do that easily in the clinical setting in a reliable way, it equips clinicians with better information when deciding on the most appropriate form of treatment for patients, be that ongoing surveillance, radiosurgery or tumour surgery.
Additionally, this tool would help with speeding up the process with radiosurgery planning, meaning that it’s standardised, quicker and because of that more patients can be treated.
The team is also working on ensuring the tool can be used for multiple diseases.
A clinician will be able pick between the disease they will look at, and there will be an algorithm associated to it. So far the algorithm has been trained on vestibular schwannoma, glioma and on foetal brain images.
The interface provides a way to adjust the segmentation that has been created so clinicians can develop an automatic segmentation tool for every single task needed.
Research student Reuben Dorent who is developing the algorithm said this is one of the novel aspects of the tool.
"There are two novel aspects, one providing vestibular schwannoma an algorithm for automatic segmentation and secondly showing the segmentation can be adjusted,” he said.
The main challenge for the team is working on generalising the tool.
“We are working on getting access to a larger data set that will cover different scans, different acquisition protocol, different images, types of images in order to have a robust algorithm – an algorithm that is able to deal with different qualities of images,” Mr Dorent said.
Mr Shapey said: “The key obstacle to wider implementation is to generalise it so that it can be used on any scanner with images obtained using any parameters, whereas the current system has been trained on a specific, very uniform data-sets of patients undergoing gamma-knife treatment."
“We’re working on ways to generalise the method. If it generalises well then we can introduce it into a workflow within a research setting, hopefully within 1-2 years; following which, we plan to conduct a prospective clinical trial to assess its effectiveness.”
