
Professor Wolfgang Maret
Professor of Metallomics
Research interests
- Biomedical and life sciences
Biography
The biochemical principles of life are based on both organic and inorganic chemistry. My laboratory focuses on the inorganic biochemical aspects, namely how the nutritionally essential transition metal ions maintain human life and how they support growth and development. Manganese, iron, copper, and zinc ions are constituents of thousands of proteins and function in enzymatic catalysis and protein structure. Transition metal ions regulate protein functions and proteins regulate their availability. For these activities, proteins employ dynamic coordination environments that link metal ion binding and protein conformational changes.
Cellular metal ion homeostasis requires multiple proteins for transport, sensing, chaperoning, and other functions in a network of tightly controlled interactions and with full integration into metabolism and signaling. The metal-regulatory proteins employ specific molecular mechanisms. One mechanism is the sulfur-ligand centered reactivity in zinc/thiolate coordination environments. Sulfur donors confer redox activity on the otherwise biologically redox-inert zinc ion. This coupling between zinc and redox metabolism provides a way of controlling zinc binding and protein functions.
Fundamental insights into the control of transition metal ion homeostasis will lead to an understanding of the pharmacological activity and toxic actions of metal ions, and aid in developing strategies for optimizing human health and for preventing, diagnosing, and treating human diseases.
Research

Metal Metabolism Research Group
The Metal Metabolism Research Group aims to understand the roles of minerals in biology, health and disease. Much of our work is mechanistically based and involves studies at the molecular and cellular levels as well as animal model systems and human. Our research focus is currently on iron and zinc.
Physiological Oxygen Laboratory
Cardiovascular - Physiological Oxygen Laboratory

King's Water Centre
Researching water, environment and development. Our centre spans the humanities, social, and physical sciences to explore the challenges of water governance from global to local scales.
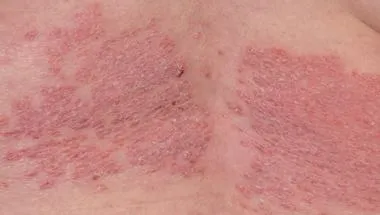
Dermatology and nutrition (nutritional dermatology)
Dermatology and nutrition (nutritional dermatology) is an emerging field that recognises the role that diet may play in the management of chronic skin disorders.

Mental health and diet
This research group aims to explroes the (1) mechanisms of food compounds, nutrients, foods, and whole diets on mental health and illness, and (2) the clinical efficacy and effectiveness of such nutritional interventions.
Immunity, nutrition and diet
Nutrition plays a critical role in immune function by providing energy and metabolites that allow metabolic reprogramming of immune cells.
Mineral nutrition and metabolism
Since trace elements are involved in virtually all biological processes, biometals research is relevant and can be applied across the areas of biomedical and health research.
Features
Our essential elements
A look through the periodic table with Professor Wolfgang Maret
Research

Metal Metabolism Research Group
The Metal Metabolism Research Group aims to understand the roles of minerals in biology, health and disease. Much of our work is mechanistically based and involves studies at the molecular and cellular levels as well as animal model systems and human. Our research focus is currently on iron and zinc.

Physiological Oxygen Laboratory
Cardiovascular - Physiological Oxygen Laboratory

King's Water Centre
Researching water, environment and development. Our centre spans the humanities, social, and physical sciences to explore the challenges of water governance from global to local scales.

Dermatology and nutrition (nutritional dermatology)
Dermatology and nutrition (nutritional dermatology) is an emerging field that recognises the role that diet may play in the management of chronic skin disorders.

Mental health and diet
This research group aims to explroes the (1) mechanisms of food compounds, nutrients, foods, and whole diets on mental health and illness, and (2) the clinical efficacy and effectiveness of such nutritional interventions.

Immunity, nutrition and diet
Nutrition plays a critical role in immune function by providing energy and metabolites that allow metabolic reprogramming of immune cells.

Mineral nutrition and metabolism
Since trace elements are involved in virtually all biological processes, biometals research is relevant and can be applied across the areas of biomedical and health research.
Features
Our essential elements
A look through the periodic table with Professor Wolfgang Maret

