The Taams lab studies fundamental cellular and molecular mechanisms that initiate, perpetuate and regulate immune-mediated inflammation in human health and disease. Our long-term goal is to identify novel targets and pathways for therapeutic intervention of inflammation and to better inform treatment decisions. To achieve this, the lab works in a multi-disciplinary environment with strong clinical, academic and industrial collaborations.
The lab has a specific interest in the interactions between T cells, monocytes and stromal cells and how their cross-talk influences inflammation in the tissue microenvironment. We also have a growing interest in the cross-talk between the immune and nervous systems, and how this relates to pain and neuronal function.
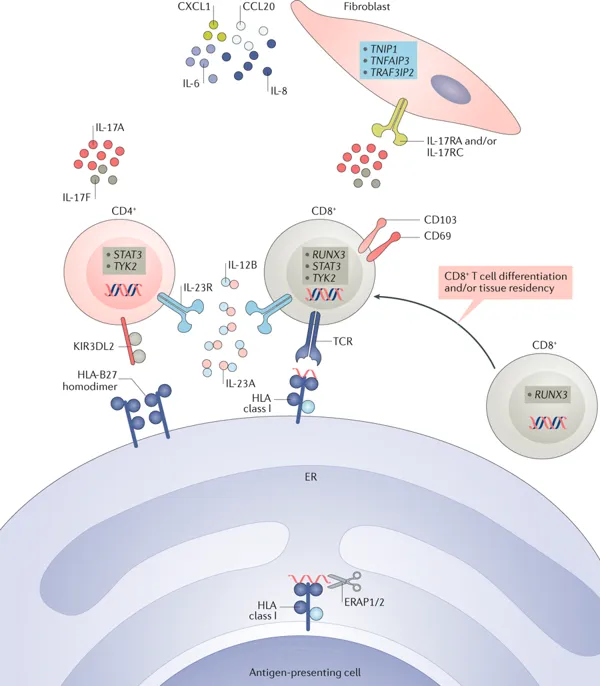
We approach our research questions through a combination of in vitro models with ex vivo and in situ studies using human cells and tissue samples to investigate the presence, function and regulation of pro-inflammatory and anti-inflammatory immune cells, and their interactions with stromal or neuronal cells. We translate our questions and findings by studying rheumatoid arthritis (RA) and psoriatic arthritis (PsA) as key examples of chronic inflammatory, painful diseases and as powerful model systems to investigate the immune system at the site of inflammation in humans.

Our Partners

Versus Arthritis

Medical Research Council

Biotechnology & Biological Sciences Research Council

IMI RTCURE

IMI BTCURE

