This landmark trial provides evidence that a faecal transplant can improve gut health by modifying the gut microbiome and reduce ammonia levels in patients with cirrhosis. Initial findings from PROFIT are promising news for patients with chronic liver disease who are in desperate need of alternative treatment options.
Chief Investigator of the PROMISE trial, Professor Debbie Shawcross from the School of Immunology & Microbial Sciences
22 June 2023
'Poo transplant' trial provides hope for liver disease patients
The UK is to launch a clinical trial of a ‘poo transplant’ that researchers believe could treat advanced liver disease and fight antimicrobial resistance.
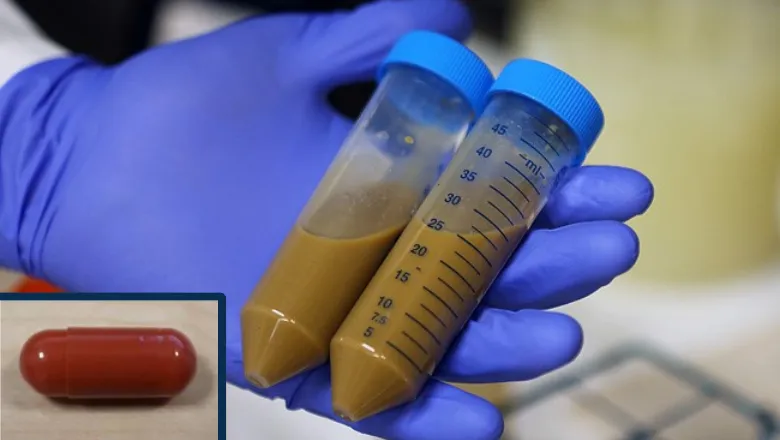
The trial’s investigators also provide evidence for the first time that a faecal transplant can dramatically improve gut health.
The PROMISE trial is led by King’s College London and funded by a National Institute for Health and Care Research (NIHR) and Medical Research Council (MRC) partnership. It will recruit patients with cirrhosis from across the UK to test whether oral Faecal Microbiota Transplant (FMT) capsules from freeze-dried stool from healthy volunteers reduces the likelihood of getting an infection.
End-stage chronic liver disease, known as cirrhosis (irreversible scarring of the liver), is the third biggest cause of mortality and loss of working life in the UK. The human body contains trillions of bacteria, but people with cirrhosis have an increased number of ‘bad’ bacteria in the bowel which makes them highly susceptible to a host of infections. Over-prescribing antibiotics means they are becoming less effective to treat these infections, and the bowel can become infected with ‘super-bugs’. Infections in people with cirrhosis are often severe and can be fatal.
Researchers from King’s College London show that the bad bowel bacteria in these patients can be replaced with healthy bacteria from healthy volunteers in a process known as FMT.
Findings from the research’s group initial FMT trial, the PROFIT trial which is also funded by the NIHR, is announced this week at the EASL 2023 Congress in Vienna, Austria.
The PROFIT trial was a 32-person safety and feasibility trial that was the first of its kind in liver patients in Europe. In this trial, FMT was administered via endoscopy. Results showed not only that it was safe and well-tolerated in these patients, but that it modifies gut microbiota, enhances intestinal barrier function and antimicrobial mucosal immunity, and boosts the metabolism of the toxin ammonia.
These significant findings bring a renewed sense of hope for patients grappling with advanced cirrhosis and offer a potential breakthrough in their treatment. The next steps are to open the PROMISE trial, where FMT will be delivered via capsules which removes the need for invasive endoscopy procedures.
She added: “We are now expanding this trial nationally to 300 patients across the UK in the PROMISE trial. Patients told us that they would prefer to take tablets rather than have an endoscopy. The ‘crapsules’, which have none of the taste or smell as the name suggests, may offer new hope for patients with cirrhosis who are out of treatment options.”
In many cases, the only definitive treatment option for cirrhosis patients is a liver transplant, however this cannot be safely performed if the patient has an antimicrobial resistant infection. Patients with cirrhosis are at particularly high risk for antimicrobial resistance because of their disproportionate exposure to antibiotics – 25% of patients are on long-term antibiotics.
Infections, especially resistant infections, are a death sentence to liver patients. Patients with chronic liver disease are often prescribed antibiotics, however, they are at high risk of multidrug-resistant infections. This is contributing to the global health crisis of antimicrobial resistance.
Dr Lindsey Edwards, from the School of Immunology & Microbial Sciences
Pamela Healy, Chief Executive of the British Liver Trust who are collaborators in the PROMISE trial said: “We are delighted to support this innovative research that could become a life-changing treatment for patients. Not only can untreatable infections be serious themselves, but they can also trigger further serious complications such as hepatic encephalopathy or ascites. The researchers have worked with the Trust to ensure that patients have been actively involved in all the stages of the study from its initial conception through to design and delivery.
“Building upon the promising results from the PROFIT trial, we are excited about the commencement of the PROMISE trial, a critical endeavour that will shed further light on the advantages of this treatment for individuals with cirrhosis who develop drug-resistant infections. This exciting new research also has wider implications – potentially in the future it could tackle antimicrobial resistance. Finding new, effective ways to treat resistant bacteria is one of the most important challenges in global medicine and this could provide a solution that could save healthcare systems across the world, millions of pounds.”
The PROMISE trial is funded by the National Institute of Health Research UK (NIHR). The trial will recruit 300 patients from up to 16 sites across the UK (including hospitals in London, Wales, Gateshead, Leeds, Newcastle, Nottingham, Derby, Liverpool, Glasgow, Dundee, Bristol, Plymouth and Portsmouth). Participants will be allocated randomly to receive either FMT capsules or placebo (an identical-looking dummy tablet which contains no FMT) every 3-months for two years.
For more information visit: https://www.fmt-trials.org/promise/faqs-2/


