Our fundamental research shows that cell migration is even more tightly controlled than anticipated, which not only helps us to understand how the human body works but also reveals potential novel drug targets to prevent cancer metastasis.”
Dr Matthias Krause
28 September 2021
Advanced microscopy study reveals molecular details of tight control of cell migration
New research provides novel insights with potentially important implications for our understanding of Nance-Horan Syndrome and cancer metastasis.
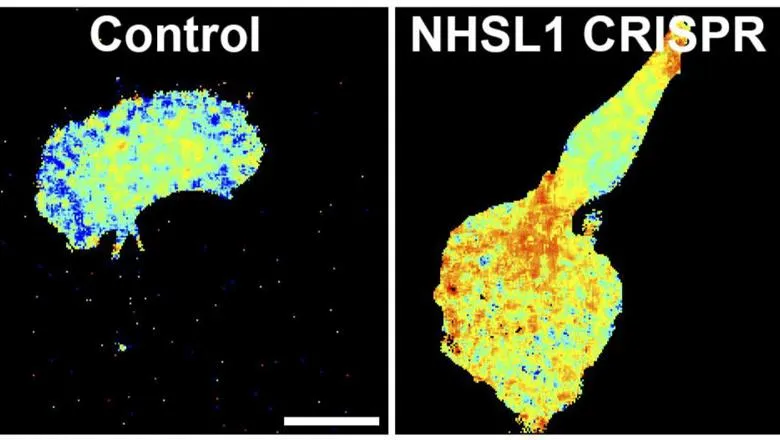
Scientists at King’s have used advanced microscopy techniques including FRET-FLIM microscopy, computational image analysis, biochemical and cell biological approaches to reveal a novel mechanism in how mammalian cells tightly control cell migration.
The research, published today in Nature Communications, is the result of collaboration between the Krause Lab and the Stramer Lab, both in the Randall Centre for Cell & Molecular Biophysics at King’s, and the Ameer-Beg Lab in the School of Cancer & Pharmaceutical Sciences.
Cell migration is fundamental for the existence of multicellular animals, including humans, as certain cells migrate throughout the embryo to form specific tissues. It is also crucial for human health throughout life – for example during acute wound healing. Its deregulation allows cancer cells to leave the primary tumour and colonise distant sites (cancer metastasis).
Even though cell migration has been studied for a long time, how it is tightly controlled is not well understood. The polymerisation of a protein (actin) network behind the plasma membrane provides the pushing force for membrane protrusion and, consequently, cell migration.
The research by the Krause, Stramer and Ameer-Beg Labs shows that this tight control of cell migration is mediated by the Nance-Horan Syndrome-like 1 (NHSL1) protein which inhibits the initiation of actin polymerisation at the leading edge of migrating cells. NHSL1 belongs to the poorly investigated Nance-Horan Syndrome protein family along with Nance-Horan Syndrome (NHS) and NHSL2 proteins. Mutations in the NHS gene cause Nance-Horan syndrome, which is characterised by dental abnormalities, developmental delay, and congenital cataracts.
The scientists are now turning to NHS to study whether deregulated cell migration may play a role in the pathogenesis of Nance-Horan Syndrome. In addition, they are investigating whether the Nance-Horan Syndrome protein family act as tumour suppressors because deregulation of cell migration is a hallmark of cancer metastasis, and NHSL1 is required for tight control of cell migration.
Read the full paper Nance-Horan Syndrome-like 1 protein negatively regulates Scar/WAVE-Arp2/3 activity and inhibits lamellipodia stability and cell migration in Nature Communications.



