Within the Department of Imaging Chemistry and Biology we have a number of projects developing contrast agents to better understand the fundamental immunology of inflammation, transplant rejection and monitoring of therapy response
In vivo tracking of labelled stem and immune cells is a key component of several projects within the department, using both reporter genes and direct ex vivo labelling of cells, in conjunction with PET, SPECT and MR. Literature methods for radiolabelling cells abound but evidence that labelled cells behave and replicate normally is sparse and contradictory. It is imperative to check cell function and survival with a range of general and specific assays and minimise toxicity by selecting delivery systems that control sub-cellular contrast agent distribution.
Imaging activated complement post ischemia reperfusion injury
Ischaemia reperfusion (I/R) injury occurs when blood flow is restored after extended ischaemia. It causes morbidity and mortality in myocardial infarction, stroke, gut ischemia, and cardiopulmonary bypass and impairs graft survival in transplanted kidney, heart and lung.
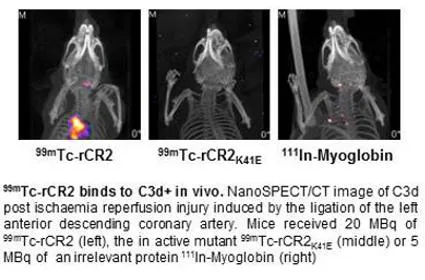
Molecular oxygen, neutrophils, and components of the activated complement (C) cascade all contribute. Recently it has been shown that complement proteins, such as C3, are synthesised by tissue parenchyma following tissue stress or infection. C3 is cleaved at the cell surface to form first the C3b fragment and then membrane anchored C3d. Together with Steven Sacks and Richard Smith in the KCL MRC Center for Transplantation we have developed an imaging agent to target C3d on the surface of cells after I/R injury in transplanted organs and on heart cells post myocardial infarction. The aim of this work is to assess whether this will measure local innate immune activity more meaningfully than measuring circulating protein. The radiolabelled conjugates will be used to image C3d in tissue damaged in vivo by ischemic reperfusion in animal models of IR injury of the heart and kidney.
Imaging cellular Immunotherapies
As well as looking at molecular imaging agents for imaging inflammation and transplant refection we are also investigating the possibility of monitoring cellular therapies. CD4+CD25+ FoxP3+Regulatory T-cells (Tregs) are critical to initiation and maintenance of autoimmunity, allergy and transplantation tolerance. It is thought that alloreactive effector CD4+ T cells drive chronic allograft rejection and that control of these cells by Tregs could achieve transplant tolerance. In vivo imaging of transplantation tolerance would be a valuable tool to track ex vivo expanded Tregs to sites of transplants, and determine the duration of their effectiveness. Retroviral transduction of Tregs with a reporter gene (hNIS) will allow imaging over days to months, using F-18 tetrafluoroborate without risk of radiation damage by high radiolabel load.